CHICAGO CLINICAL RESEARCH INSTITUTE
Multi-specialty Group Practice And Dedicated Research Site Conducting Phase I To Phase Iv Clinical Trials.
20+
Specialists
4600+
Patients Benefitted
350+
Clincial Trials Completed
70+
Appointments Scheduled/Week
BEGIN YOUR JOURNEY IN CLINICAL TRIALS

Vaccine Trials Currently Enrolling Participants. Evaluating Immune Response, And Side Effects.

Clinical Research For Gout Relief. Less Inflammation.
Healthier Joints.

Advancing Sjögren's Research. Reducing Overactivity.
Restoring Quality Of Life.

Developing Effective Therapeutic Solutions For Type 2 Diabetes Mellitus(T2DM).

Clinical Study On Biologic Therapies To Lower LDL-C.
Improve Long-Term Health.


Accurate Lupus Diagnosis.
Early Intervention.
Better Outcomes.
OUR PRINCIPAL INVESTIGATORS

Dr. Azaz Ahmed MD, FACP
Internal Medicine

Dr. Norman M.James MD, FACP, FACR
Board Certified in Internal Medicine

Ann Kuehl MSPAS, BS
PA-C, Dermatology
50+
Years of Experience
Meet Our Clinical Expert
Dr.Levinson MD, FACP, FACR
Expertise: Rheumatology
Advocate Illinois Masonic Medical Center and Advocate Trinity Hospital. He received his medical degree from Chicago Medical School/Rosalind Franklin University of Medicine Science and has been in practice for more than 40 years. He has expertise in treating diabetes, hypertension & high blood pressure, spinal stenosis, lupus, gout and spondylosis.
WHY CHOOSE US FOR YOUR CLININCAL TRIALS
CCRII has a combined 70 years of clinical research experience and has conducted more than 350 clinical research trials in the therapeutic areas of Diabetes Mellitus Type II, Hypertension, Hyper-lipidemia, Chronic Kidney Disease, Cardiovascular Disease, Osteoarthritis, Rheumatoid Arthritis, Pain Management, Neuropathy, Gynecology, Male Health, Ophthalmology, and Oncology.
We are a Phase I-IV research facility and we have conducted over 350 research studies to date. Through a dedicated and experienced team, we provide the utmost quality of care to all of our research participants.

70+
Years
Experienced

Make An Appointment
HOW TRIALS WORK

Schedule A Pre-Screening Call
This call involves screening over the phone to ensure we have the right trial for you.

Get Qualified For A Trial
Medical coordinators will determine which therapetic area would good fit.

Schedule an Appointment For Your First Visit.
You get enrolled into a trial, get briefed on the specifics of the trial and corresponding trial visits.
FREQUENTLY ASKED QUESTIONS
When considering participating in a clinical trial, it is important to make an informed decision. Below are answers to frequently asked questions that many potential volunteers have about participating in a study.
What is a clinical trial?
A clinical trial is a research study in which volunteers receive investigational treatments under the supervision of a physician and other research professionals. These treatments are developed by pharmaceutical and biotechnology companies who select qualified physicians, also known as investigators, to conduct clinical trials to determine the benefits of investigational drugs.
Clinical trials are usually conducted in three phases (I, II, III). Only a small number of people participate in phase I trials while the later phases involve a larger number of volunteers.
Who can participate in a clinical trial?
All clinical trials have guidelines about who can participate. Before joining a clinical trial, a volunteer must qualify for the study. The factors that allow volunteers to participate in a clinical trial are called “inclusion criteria” and the factors that disallow volunteers from participating are called “exclusion criteria.” These criteria can include age, gender, the type and stage of a disease, previous treatment history, and other medical conditions.
Some research studies seek participants with specific illnesses or conditions to be studied in a clinical trial, while others require healthy participants. It is important to note that inclusion and exclusion criteria are used to identify appropriate participants, promote participants’ safety, and ensure that researchers learn the information they need.
How does a clinical trial work?
In a clinical trial, a volunteer is usually assigned a specific study group. Volunteers in one study group may receive an investigational treatment or study drug while other volunteers may receive a placebo or a treatment already available.
A placebo is an inactive product used to assess the experimental treatment’s effectiveness. The participant, physician, and research staff may not know which volunteer receives a placebo and which receives the active treatment. Not knowing which participants are receiving the active treatment allows the physician and research staff to objectively observe the volunteers during the study. Regardless of which treatment volunteers receive, however, the level of medical attention and care that each receives is the same.
What happens after the trial?
After a study phase is complete, the data is collected to determine the drug’s effectiveness, if it is safe and if there are any side effects. Depending on the results, researchers then determine whether to stop testing or move to the next phase of study. After phase III of a study is complete, researchers decide if the results are medically important and may submit them to journals for peer-review.
Data then may be submitted to the Food and Drug Administration (FDA) for approval. If a drug is approved, pharmaceutical companies may continue to conduct studies that compare the new drug—in terms of its safety, effectiveness, and cost—to other drugs already on the market or assess a drug’s long-term effectiveness and its impact on the quality of a person’s life.

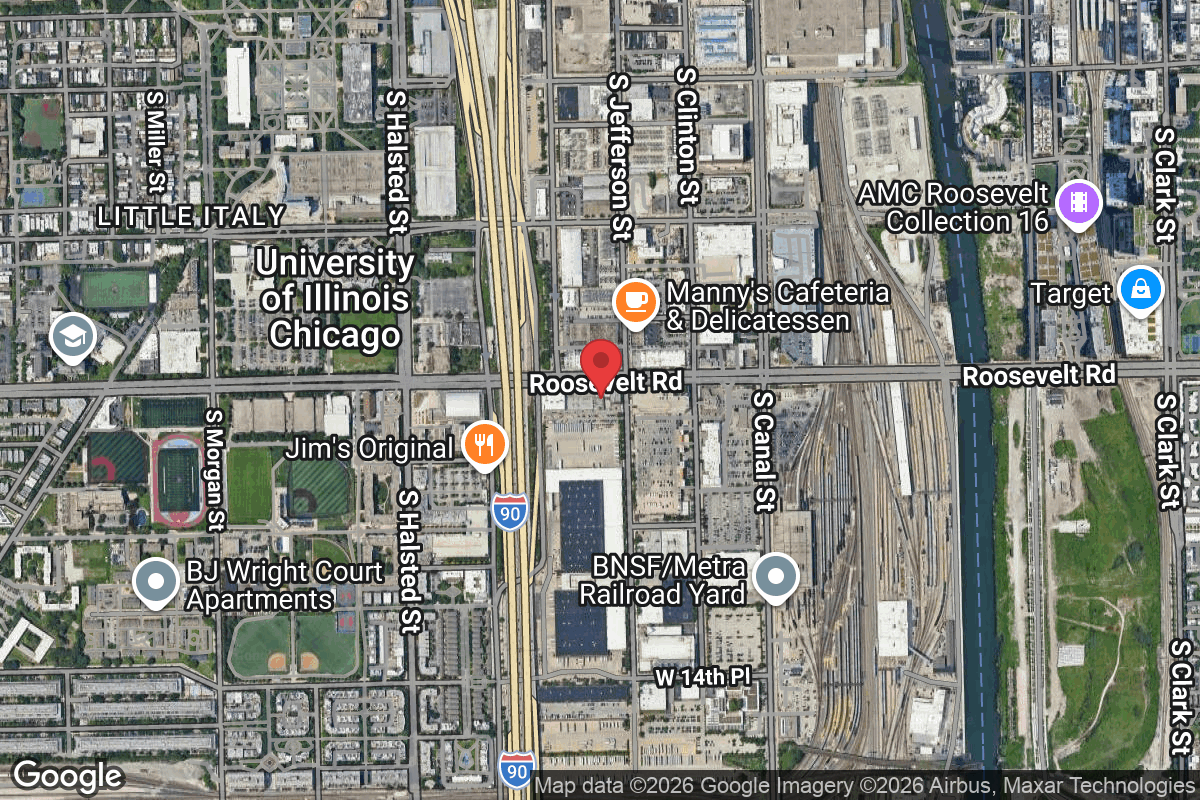
Address
611 W Roosevelt Rd, Chicago IL 60607 USA

Call Us
(312) 823 - 6800

Send us a Mail

Clinic Hours
Mon-Fri: 8:00 AM - 5:00 PM
Get in Touch with us
We’re here to help anytime

Call Us

Mail Us

Opening Time
Mon -Sat: 7:00 - 17:00